Animals in science have multiple layers of oversight ranging from the federal government that creates laws and regulations, numerous committees, to day to day animal care staff and even the court of public opinion. All of these layers play a role in oversight of animals involved in medical research.
Laws Protecting Animals
Understanding the protections in place.
Layers of Oversight
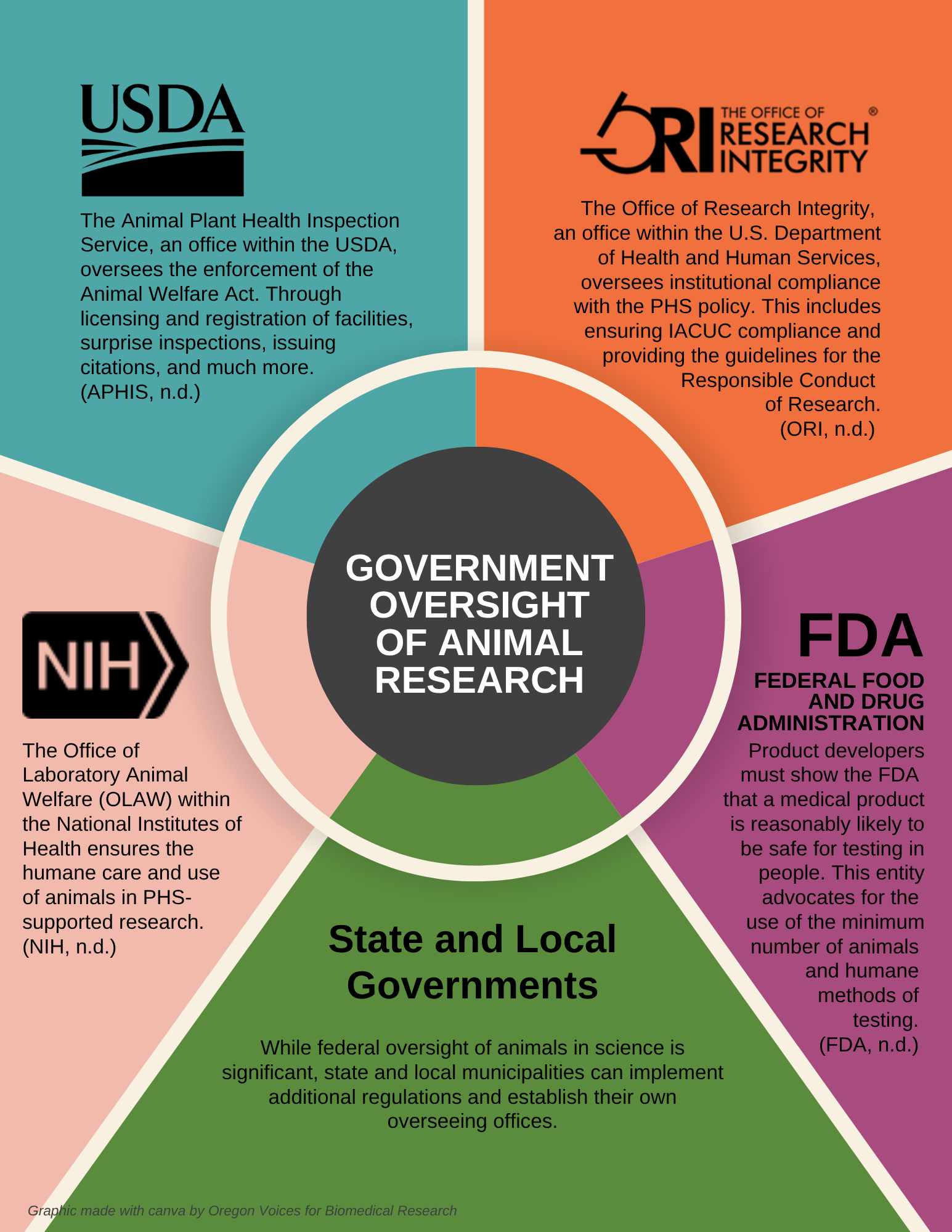
Government Oversight
Animal Welfare Act & Regulations (AWRs) provide the basis for most animal protection in medical research (also zoos, breeders, etc.). Although rats and mice make up the majority of animals in medical research, they are actually exempted from the AWRs. These regulations are enforced by the United States Department of Agriculture (USDA) that performs 1-2 unannounced inspections per year at all registered research facilities. They have a zero-tolerance policy for any deviation from the AWRs. Facilities will receive citations even if they self-report accidents, mistakes or adverse events as soon as they occur.
Public Health Service (PHS) Policy requires all facilities that enroll any vertebrate animals (including fish, rats and mice that are exempted from the AWRs) in medical research to abide by The Guide for the Care and Use of Laboratory Animals. The PHS policy is overseen by the Office of Laboratory Animal Welfare (OLAW) and any facility receiving government funding, for example from the National Institutes of Health (NIH), must follow the policy requirements or risk having their research funding revoked.
Committee Oversight
Numerous committees oversee animals enrolled in medical research studies. Before a study is even funded, a scientific panel of experts reviews it to discuss scientific importance, relation to human health, techniques or animal species involved and decides if it meets the necessary requirements to receive funding. Once a study makes it over this hurdle, it then must undergo review by the internal research facility’s Institutional Animal Care & Use Committee (IACUC) where another panel of scientists, veterinarians, and non-scientists evaluate it for potential alternatives to animals (like NAMs) if available and general animal procedures and care. In addition to USDA unannounced inspections, the IACUC performs semiannual facility inspections required by AWRs and PHS Policy. Scientists also have to provide annual updates as the study progresses and report any unexpected adverse events. Depending on the type of event, these may also require notification to the Office of Laboratory Animal Welfare. The IACUC has the power to suspend any study if necessary.
Additional committees that review animal care include accrediting organizations that perform inspections of all facilities seeking gold standard accreditation. Similar to pet hospitals that can be accredited by the American Animal Hospital Association, human hospitals accredited by the Joint Commission, and Zoos accredited by the Association of Zoos & Aquariums; research facilities can seek gold standard accreditation by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALACi). This accreditation is strongly encouraged by OLAW and funding agencies.

Veterinary & Caregiver Oversight
Day to day animal care often includes everyone from husbandry technicians, to animal nurses, to study staff and veterinarians. Animal care staff are the most direct advocates for animals in managed care. They provide daily health checks, feeding, cleaning, snacks, medications, socialization and training. They know and care about each individual animal, and are powerful advocates. Their opinions are taken seriously by everyone, and they can report to the IACUC or the Attending Veterinarian any animal related concerns. Veterinary oversight is required by law in research facilities, unlike for companion pets. Additionally companion pets may see their veterinarian 1-2x per year, but research animals are often seen multiple times per week by veterinary staff. All veterinarians take an oath for the protection of animal health and welfare, the prevention and relief of animal suffering, the conservation of animal resources, the promotion of public health, and the advancement of medical knowledge. The veterinarians who care for research animals are uniquely situated to fulfill these responsibilities by caring for animals and guiding life-saving science.
 Photo: Americans for Medical Progress via comeseeourworld.org
Photo: Americans for Medical Progress via comeseeourworld.org Public Oversight
Public perception of animals has long shaped how they are treated and regulated—not only in medical research, but also in zoos, pet stores, agriculture, and even dog fighting survivors to name a few. This relationship has been a decades-long push and pull. Today, information can spread across social media within minutes whether accurate, misleading, or somewhere in between, and it can powerfully influence public opinion.
Lab animals matter. They matter to the veterinarians and staff who care for them, to the scientists who work with them, to the doctors who turn discoveries into treatments, and to the patients whose lives depend on that work. They should matter to all of us. At the same time, it’s essential to evaluate information critically in an environment where emotionally charged language can distort understanding. For example, one account might claim that “babies were ripped from the womb,” while another describes the same event as a “routine C-section”. These two phrases evoke very different reactions.
Before forming conclusions, individuals should seek out accurate, up-to-date information from credible sources. Ask thoughtful questions, examine potential biases, and look for evidence-based explanations. As in any field, there are instances of misconduct, and those must be addressed to uphold ethical standards in animal care and research.
Animal health, physical and psychological, is extremely important to maintain the scientific discoveries that benefit you, your relatives, and future generations. Every animal involved in research plays an important role, and a well-informed public is key to understanding and valuing those contributions, including those “routine c-section” animals that are helping our human NICU infants have better health outcomes.

Ethical Oversight at Every Step
Every research procedure follows strict ethical protocols designed to minimize impact while advancing discoveries that save lives.
IACUC Review
Every procedure undergoes rigorous review by Institutional Animal Care and Use Committees, which include veterinarians, scientists, ethicists, and community representatives.
Pain Management
Anesthesia protocols, pain management strategies, and post-operative monitoring are developed collaboratively between veterinary specialists and research teams to ensure the highest welfare standards.
Continuous Improvement
The commitment to humane science drives continuous improvement in techniques and technologies, reducing the impact on animals while advancing discoveries that benefit both human and animal health.